Special Feature: Hospital Mortality and ICU-Acquired Infection
Special Feature
Hospital Mortality and ICU-Acquired Infection
By Jun Takezawa, MD
Risk Factors for the ICU-Acquired Infection
Several factors are considered to be associated with the development of nosocomial infections in the ICU (see Table 1, below). Among them, indwelling devices that directly contact the blood and mucosal membrane such as the central venous catheter, urinary tract catheter and endotracheal tube are considered to be the most responsible risk factors in the development of nosocomial infections. These devices are placed into the patient and manipulated by the medical practitioner, and referred to as external risk factors. These device-related external risk factors are associated with the length of time the device remains in the patient. However, they are also associated with the frequency of manipulations of the device, such as bolus injection and exchanges of the infusion bottles and lines, especially for indwelling central venous catheters. In addition to the length and/or frequency of exposure to the risk device, the hygienic management, behavior pattern of antibiotic administration, level of infection control, and patient management (therapeutic, nursing, monitoring, staffing, and organizational) also play a role, along with the external risk factors, in the development of nosocomial infections. On the other hand, the risk factor inherent to the patient is referred as an internal risk factor. Such internal risk factors include age, gender, severity of illness, immunological competence, comorbidity, and so on.

In order to accomplish an inter-institutional comparison on infection rate, both internal and external risk factors should be adjusted. Among the risk factors indicated above, the internal risk may be adjusted by using measures of illness severity such as the APACHE score, but the external risk can only be adjusted by device utilization days. Therefore, the difference in infection rates adjusted by the above two risk factors is attributable to the other remaining external risk factors, most of which are related to both the patient and ICU management.
Purpose of Surveillance
The purpose of the surveillance is 1) to identify the outbreak of nosocomial infections (although outbreaks are usually readily noticed by ICU practitioners); 2) to provide data on infection control to be pursued by ICU practitioners in quality improvement; 3) to obtain the incidence and prevalence of nosocomial infections from the viewpoint of public health; and 4) to provide for inter-institutional comparisons with respect to preventive programs and practice in managing nosocomial infections by the respective institutions.
When surveillance is conducted for the purpose of inter-institutional comparison of the nosocomial infection rate, all risk factors for ICU-acquired infections should be adjusted. The National Nosocomial Infection Surveillance (NNIS) system, which is run by the US Centers for Disease Control and Prevention (CDC), apparently uses only external risk-adjusted infection rates for inter-institutional comparison. The severity of illness in NNIS employs the device utilization ratio, which is calculated as the length of days the devices are in use divided by the number of patient days. Use of this ratio is based on the assumption that the severely ill patient requires long-term use of the devices for efficient and safer management. However, the device utilization ratio, as well as APACHE and SAPS scoring systems, which are frequently used for stratifying severity of illness in terms of mortality, are not proved to be related to the acquisition of nosocomial infections in the ICU, in part because the most severely ill patients die quickly. Therefore, patients who die within 24 hours after admission to the ICU are excluded for inter-hospital comparison of the performance of ICUs.
In the NNIS system, risk-adjusted infection rate is compared within the individual types of ICUs, such as neuro-ICU, coronary-CU, and surgical-ICU, which implies that the original disease is taken into account as an internal risk factor. However, because all the internal risk factors are not included in the NNIS system, the exact effect of ICU-acquired infections on hospital mortality is unknown.
ICU-Acquired Infection and Hospital Mortality
Although the incidence of ICU-acquired infection is recognized as an important determinant of outcome for ICU patients, the precise relationship between ICU-acquired infection and hospital mortality has yet to be defined. A 1-day point-prevalence study for 1417 ICUs from 17 western European countries, called the EPIC study, showed that a prevalence rate of infection in ICUs was 44.8%, and almost half of the infections were acquired in the ICU (20.6%).1 The EPIC study showed that the impact of ICU-acquired infection on ICU mortality might vary according to the types of infection; the highest odds ratio was found in sepsis (3.50), followed by pneumonia (1.91) and blood stream infection (1.73). Moreover, several studies showed that inadequate treatment of infections might be an important determinant of hospital mortality.2,3
There have been few cohort studies in which the patients discharged from the ICU were followed up until hospital discharge. One cohort study involving 28 ICUs from 8 countries showed that the hospital mortality rate in patients with ICU-acquired infection was 32.1%, compared with 12.1% in patients without ICU-acquired infections.4 These rates were crude and not adjusted for potential confounders (eg, age, underlying disease, and severity of illness).5,6 Moreover, the impact of ICU-acquired infection on hospital mortality might be affected by drug-resistant pathogens.7
JANIS Database Analysis
The Japanese Nosocomial Infection Surveillance (JANIS) system, started in 2000 by the Ministry of Health, Labor, and Welfare, collected data on 7374 patients admitted to the 34 participating ICUs between July 2000 and May 2002. The data used for their analysis is from patients discharged from ICU who were aged 16 years or older, whose ICU stay was from 48 to 1000 hours, who had not transferred to another ICU, and who had no infection diagnosed within 2 days after ICU admission. These patients were followed up until hospital discharge or the 180th day after ICU discharge. Adjusted hazard ratios (HRs) with their 95% confidence intervals (CIs) for hospital mortality were calculated using a Cox’s proportional hazard model.8
Table 2 shows the effect of ICU-acquired infections on hospital mortality in the JANIS data. Overall, 678 patients (9.2%) had at least one ICU-acquired infection. Drug-resistant pathogens were detected in 201 patients. The most common ICU-acquired infections were ventilator-associated pneumonia (VAP, 517 cases, 64%), followed by sepsis (106 cases, 13%), surgical site infections (102 cases, 13%), urinary catheter-related infections (43 cases, 5%), and catheter-related blood stream infections (42 cases, 5%). All types of ICU-acquired infections were significantly associated with hospital mortality. Compared to patients who had no infection, those infected by drug-susceptible and drug-resistant pathogens had significantly higher rates of hospital mortality (shown as P value). The mortality rate with drug-resistant pathogens was higher than that with drug-susceptible pathogens, except for urinary tract infection in which few cases of drug-resistant pathogens were observed (not shown here).
Table 3 shows hazard ratios and their corresponding 95% confidence intervals for hospital mortality. After adjusting for sex, age, and APACHE II score, significantly higher HR for hospital mortality was found in respirator, central venous catheter, and ICU-acquired infection caused by drug-resistant pathogens, with significantly lower HR for elective and urgent operation and urinary catheter. The impact of ICU-acquired infection on hospital mortality was different between drug-sensitive pathogens (HR, 1.11; 95% CI, 0.94-1.31) and drug-resistant pathogens (HR, 1.42; 95% CI, 1.15-1.77).
Severity of Illness and ICU-Acquired Infection
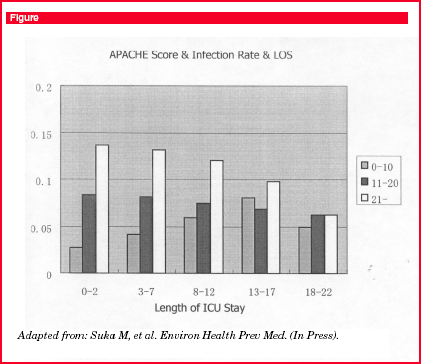
It is still unknown whether severity of illness is related to the development of ICU-acquired infections. When the incidence of ICU-acquired infections is evaluated in terms of severity of illness along with the ICU stay, the incidence of ICU-acquired infections along the ICU days is different among the severity of illness (see Figure, below ).8 In the most severely ill patients, the incidence of ICU-acquired infections is highest in the early phase of ICU admission, while in the least severely ill patients, the incidence of ICU-acquired infections is low in the early phase, but is increased along the ICU stay up to 20 days. In moderately ill patients, the incidence ICU-acquired infections do not change markedly along the ICU stay. Therefore, severity affects the incidence ICU-acquired infections; however, this effect on ICU-acquired infections is inversed depending on the severity of illness. In this sense, the general concept that the more severely ill the patients are, the more they develop nosocomial infections is not verified.
Performance Measurement of ICUS
Performance of the ICU is usually measured in terms of outcome and process. The incidence of ICU-acquired infection is classified as the process evaluation, while hospital mortality is classified as outcome evaluation. However, the sensitivity of the outcome measurement by hospital mortality is low, because the relatively small numbers of the patients die during the hospital admission. Additionally, so many confounders are associated with the hospital mortality of ICU patients, which include original disease, severity of illness, development of complications (medical errors and nosocomial infections), patient management (therapeutic, nursing and monitoring capabilities), demographical characteristics (age and gender of the patients), and organizational characteristics (open or closed ICU, staffing). Because the magnitude of contribution of those confounders on mortality is not prioritized, it is extremely difficult to evaluate ICU performance on an individual confounder (risk factor) basis. It is of most importance to develop a new statistical model to measure both overall and individual confounder-based performance of the ICU. The ICU-acquired infection is one of the most important confounders (risk factors) for the measurement of ICU performance. It is concluded that performance of the ICU is improved by improving the individual risk factors; however, it is extremely difficult to achieve it by just monitoring the overall risk-adjusted hospital mortality of the patients discharged from the ICU.
References
1. Vincent J, et al. JAMA. 1995;274:639-644.
2. Kollef MH, et al. Chest. 1999;115:462-474.
3. Zaidi M, et al. Arch Med Res. 2002;33:290-294.
4. Alberti C, et al. Intensive Care Med. 2002;28:108-121.
5. Jencks SF, et al. JAMA. 1988;260:3611-3616.
6. Green J, et al. JAMA. 1990;263:241-246.
7. Niederman MS. Crit Care Med. 2001;29(Suppl): N114-N120.
8. Suka M, et al. Environ Health Prev Med. (In press).
Jun Takezawa, MD, Director of Emergency and Intensive Care Medicine Professor, Department of Emergency Medicine, Nagoya University School of Medicine, Nagoya, Japan is an Associate Editor of Critical Care Alert.
Several factors are considered to be associated with the development of nosocomial infections in the ICU. Among them, indwelling devices that directly contact the blood and mucosal membrane such as the central venous catheter, urinary tract catheter and endotracheal tube are considered to be the most responsible risk factors in the development of nosocomial infections.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
