Special Feature: Update on Hemodynamic Monitoring in 2012
Special Feature
Update on Hemodynamic Monitoring in 2012
By Richard J. Wall, MD, MPH, Pulmonary Critical Care & Sleep Disorders Medicine, Southlake Clinic, Valley Medical Center, Renton, WA, is Associate Editor for Critical Care Alert.
Dr. Wall reports no financial relationships relevant to this field of study.
Hemodynamic monitoring is an essential part of caring for critically ill patients. Critical care providers are regularly faced with the challenge of determining whether a patient is adequately volume resuscitated, and hemodynamic assessments are often the first step in making a proper diagnosis so that other life-saving therapies can be promptly implemented. The purpose of this review is to provide an overview of available hemodynamic monitoring systems, and offer general principles to guide providers who are trying to choose the best system for their particular setting.
All of us know that initial uncertainty when an obese, hypotensive patient with renal failure and/or cardiomyopathy rolls into the unit. Do they need more fluid? Are they bleeding? Could it be tamponade or a pulmonary embolism? Should we check central venous pressure (CVP) or place an arterial line? What about a pulmonary artery (PA) catheter? Should we try the newer less invasive cardiac output (CO) monitors? Would measurement of mixed venous oxygen saturation (SvO2) help, and should it be done continuously or intermittently? What about bedside ultrasound? How about another fluid challenge or vasopressors?
CARDIAC OUTPUT HEMODYNAMIC MONITORING DEVICES
While microcirculatory changes are thought to be the main culprits of organ dysfunction in shock, the reality is that technologies for monitoring microcirculation are not readily available. This disconnect has resulted in a myriad of CO (i.e., macrocirculation) monitoring devices. The choices are overwhelming. While a detailed review of every system is beyond the scope of this article, I will briefly discuss the most popular types of systems. I will also discuss two commonly used CO surrogates: CVP and SvO2.
PULMONARY ARTERY CATHETER
The PA catheter has long been the bedside "gold standard" for hemodynamic monitoring. It relies on the thermodilution technique, wherein downstream temperature changes are used to calculate CO. The advantage of the PA catheter is that it also allows measurement of CVP, PA pressure, systemic vascular resistance, cardiac filling pressures, and SvO2.
Despite an incredible array of data, studies have failed to show that placing a PA catheter improves patient outcomes.1,2 In addition, key information gleaned from the PA, namely CVP and filling pressures, do not adequately predict fluid responsiveness in hemodynamically unstable patients. Moreover, these devices are occasionally associated with serious complications including pneumothorax, arrhythmias, heart block, and PA rupture/infarction. For all of these reasons, use of PA catheters has dramatically waned in recent years. Nonetheless, most other CO monitoring devices still use the PA catheter as their reference validation standard.
LESS INVASIVE MONITORING SYSTEMS
Several new less invasive monitoring systems are available as alternatives to the PA catheter. These include the LiDCO™ Plus, PiCCO™, COstatus™, Vigileo™, MostCare™, and Flo Trac™ systems. Each system uses arterial analyses to continuously measure pulse pressure variation (PPV) and stroke volume variation (SVV). Each requires some sort of arterial line, and most also require a central venous line for calibration.
These systems use basic principles of dilution to estimate CO, similar to the PA catheter. The PiCCO uses injections of ice-cold intravenous fluids as the indicator, measuring downstream temperature changes. The LiDCO uses trace amounts of lithium chloride as the indicator, using a lithium-sensing electrode. The COstatus system calculates CO using ultrasound technology to measures changes in blood velocity following an injection of warm saline.
These new technologies have limitations. Although PPV and SVV are decent tools for predicting fluid responsiveness,3 there are no studies showing these new systems save lives or reduce length of stay. Furthermore, these systems are limited by factors that alter arterial waveform such as atrial arrhythmias, severe atherosclerosis, and aortic regurgitation. The Vigileo and MostCare systems do not calibrate via a central venous catheter but instead rely on proprietary software and make different assumptions to accomplish this goal. As a result, data cannot be superimposed from one system to another. Finally, PPV and SVV have only been validated for predicting fluid responsiveness in mechanically ventilated patients who are not initiating breaths on their own. A prior review discusses several of these issues in greater detail.4
ECHOCARDIOGRAPHY AND ULTRASOUND
At the bedside, echocardiography is a useful diagnostic tool because it can visualize cardiac chambers, valves, pericardium, and overall cardiac function. In turn, these images can guide therapy. For example, small ventricles might prompt a fluid challenge. A poorly contractile myocardium might prompt a trial of dobutamine. A dilated right ventricle would suggest pulmonary embolism or inferior infarction. Valvulopathy and tamponade can be quickly determined.
Echocardiography is also helpful because it uses Doppler-based methods to calculate CO. This is achieved by directing the ultrasound beam along the aorta. The moving red blood cells create a resultant Doppler frequency that can be used to measure flow velocity and volume, and thereby CO.
Several trends are escalating the use of bedside ultrasound in the intensive care unit (ICU). First, general ultrasound skills are now being taught in critical care fellowships, and intensivists are increasingly expected to have basic bedside ultrasound skills. Second, technologic advances have made bedside ultrasound devices affordable and portable. As a result, repeated assessments throughout the day are no longer an unrealistic goal.5,6 In the coming decade, I suspect that bedside ultrasound will become nearly as ubiquitous as the stethoscope in the ICU. An explosion of educational courses and online offerings are available.
Like every technology, echocardiography has limitations. Although fairly accurate for CO calculations, the technology requires training and is operator-dependent. In addition, echocardiography can only provide a CO snapshot. Serial and/or continuous measurements will require the clinician to spend longer amounts of time at the bedside. Furthermore, transthoracic techniques do not always yield good images and the effort/risk of transesophageal echocardiography restricts its availability.
MIXED VENOUS OXYGEN SATURATION
SvO2 reflects the balance between oxygen delivery and oxygen consumption (VO2). It depends on arterial blood saturation (SaO2), the balance between VO2 and CO, and hemoglobin (Hgb) levels. It can be explained by the Fick equation:
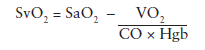
Looking at the equation, a few simple observations can be made. If CO increases and everything stays the same, then SvO2 increases. If increased VO2 is not compensated by increased CO, then SvO2 will drop. If SaO2 increases (e.g., higher FiO2 on the ventilator), then SvO2 will increase. If tissues are not capable of extracting oxygen (e.g., in the case of cell death), then SvO2 may remain high.
A true SvO2 must be drawn from the PA. Since PA catheters have become unpopular, most providers simply opt for a subclavian or internal jugular central venous catheter with its tip in the superior vena cava. Such a sample is called a central venous oxygen saturation (ScvO2). The two values are not the same and there is debate about their interchangeability.7 In addition, the relationship between CO and venous saturations is imperfect.8
Despite these limitations, monitoring of the ScvO2 is often used during resuscitation of patients with septic shock. In early goal-directed resuscitation of septic patients, there was an association between higher ScvO2 values and lower mortality.9 The Surviving Sepsis Campaign suggests a target SvO2 ≥ 70% or ScvO2 ≥ 65%.10 Real-time ScvO2 values can be continuously displayed using a specialized catheter that hooks up to the bedside monitor. No study has systematically compared continuous vs intermittent measurements of SvO2/ScvO2.
A high SvO2 is not always a good thing. It does not always signify adequate perfusion. Indeed, a high SvO2 may represent maldistrubution of peripheral blood flow and/or impaired oxygen extraction in the tissues. In septic shock, patients often have high CO and low oxygen extraction capabilities, resulting in a high SvO2. Such patients often respond favorably to additional fluid, even though their mixed venous saturations are normal-to-high.11
In summary, the SvO2 and ScvO2 are useful but imperfect indicators of cardiac output. Low venous saturations are an important warning sign for inadequate oxygen delivery. Low values should prompt the clinician to search for an underlying metabolic or circulatory impairment. The SvO2 and ScvO2 are probably most useful during the early phases of disease and resuscitation.
BIOIMPEDANCE AND BIOREACTANCE
Bioimpedance describes the response of a living organism to an externally applied electric current. It refers to the opposition to flow of that current through tissues (i.e., the opposite of conductivity). Bioreactance is an extension of this concept. It measures the change in frequency of current traversing tissue, rather than changes in impedance.
These techniques are simple to initiate, safe and non-invasive. Electrodes are placed on the patient's body, and a high-frequency, low-magnitude alternating current passes through the thorax. The impedance of this current changes as blood flow varies during the cardiac cycle. For several decades, bioimpedance technology has been used to measure CO in the perioperative setting.
Unfortunately, the technology is not reliable in critically ill patients.12 Electrical conductivity varies when patients spontaneously breathe. In addition, conditions such as obesity and chronic obstructive pulmonary disease cause changes in thoracic and/or vascular geometry that introduce error. For these and other reasons, bioimpedance has little utility in the ICU.
GENERAL PRINCIPLES OF HEMODYNAMIC MONITORING
If you are hoping to discover a perfect hemodynamic monitoring technology, I have bad news — it doesn't exist. Despite seductive marketing and high prices, every hemodynamic monitor is imperfect. In addition to scrutinizing the actual technologies, providers need to consider some principles inherent to using these imperfect tools (see Table). A recent review discusses several of these principles in greater detail.13
Table. General Principles When Using Hemodynamic Monitors
(Table adapted from Vincent JL, et al.13) |
1. Devices don't save lives. A medical device can't improve outcomes unless it is coupled with a treatment that also improves outcomes. In other words, if a hemodynamic monitoring device doesn't change behavior, it will not save lives. The data from the device must be accurate, readily available, interpretable, relevant, and responsive to change. The clinician must know how to properly apply a life-saving therapy based on the data.
2. Monitoring requirements will change during an illness. The initial monitoring system(s) must be selected based on the patient's specific needs. In many patients, an invasive approach (i.e., central venous catheter and arterial line) are needed. Once a patient stabilizes, less-invasive monitoring can be employed. Although studies failed to show a benefit for routine placement of PA catheters in the resuscitation of all critically ill patients, the PA catheter still has a role in managing selected complex cases.
3. Every patient will have different hemodynamic goals. It is unwise to universally resuscitate every patient with septic shock to a systolic pressure of 90 mmHg or CVP of 8-12 mmHg. The optimal hemodynamic targets will vary depending on age and comorbidities. Consider multiple factors in addition to the hemodynamic monitor's data. If the CVP remains low in a patient that you think is adequately resuscitated, don't reflexively give more fluid.
4. Don't rely on just one variable. Combine and integrate all available information. Use multiple monitors and tools. The differential diagnosis for a hypotensive patient with low CO is very different than for a hypotensive patient with high CO.
5. A high CO and high SvO2 are not always the best goals. Excessive administration of fluids can result in volume overload and edema, whereas excessive pressors can be detrimental in patients with coronary disease. Although Rivers noted an association between higher SvO2 and lower mortality in sepsis patients,9 that was a single-center study and their finding may not be reproducible in other settings or populations.
6. Monitor hemodynamic changes over short periods of time. For critically ill patients, there is usually more value in monitoring acute CO changes after administration of a therapy. Comparing slow changes in CO over several days is less relevant in most non-cardiac patients.
7. The cardiac output is estimated, not measured. No bedside monitor actually measures CO. For this reason, agreement of values obtained with different devices is relatively poor. The accuracy of absolute values is less important when following trends, however. A slightly less accurate value obtained by a less-invasive technique may be preferable if it can be obtained more rapidly and easily.
8. Continuous measurement is preferable. Although there is no study proving that continuous CO monitoring is superior to intermittent monitoring, continuous systems make more sense. We already routinely monitor numerous other hemodynamic variables in a continuous manner. Systems with intermittent data or requiring constant recalibration may not provide the real-time data needed for optimal management of critically ill patients.
9. Non-invasiveness is not the only goal. I agree with a prior review that recommends caution when implementing newer less invasive systems.4 Use the proper tool at the proper time to answer the proper question.
CONCLUSION
The number of choices for hemodynamic monitoring devices continues to grow. Every device has limitations, and a "perfect device" will never exist. If able, use a combination of devices, both invasive and non-invasive. Be flexible in your approach and reassess complex patients repeatedly throughout the day. When different data contradict each other, fall back on your training. Trust your intuition.
REFERENCES
- Shah MR, et al. Impact of the pulmonary artery catheter in critically ill patients: Meta-analysis of randomized clinical trials. JAMA 2005;294:1664-1670.
- Wheeler AP, et al. Pulmonary-artery versus central venous catheter to guide treatment of acute lung injury. N Engl J Med 2006;354:2213-2224.
- Pinsky MR. Hemodynamic evaluation and monitoring in the ICU. Chest 2007;132:2020-2029.
- Luks AM. Less invasive hemodynamic monitoring. Critical Care Alert 2010;17:21-24.
- Field LC, et al. Echocardiography in the intensive care unit. Semin Cardiothorac Vasc Anesth 2011;15:25-39.
- Melamed R, et al. Assessment of left ventricular function by intensivists using hand-held echocardiography. Chest 2009;135:1416-1420.
- Varpula M, et al. Mixed venous oxygen saturation cannot be estimated by central venous oxygen saturation in septic shock. Intensive Care Med 2006;32:1336-1343.
- van Beest P, et al. Clinical review: Use of venous oxygen saturations as a goal — a yet unfinished puzzle. Crit Care 2011;15:232.
- Rivers E, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 2001;345:1368-1377.
- Dellinger RP, et al. Surviving Sepsis Campaign: International guidelines for management of severe sepsis and septic shock: 2008. Crit Care Med 2008;36:296-327.
- Velissaris D, et al. High mixed venous oxygen saturation levels do not exclude fluid responsiveness in critically ill septic patients. Crit Care 2011;15:R177.
- Squara P, et al. Noninvasive cardiac output monitoring (NICOM): A clinical validation. Intensive Care Med 2007;33: 1191-1194.
- Vincent JL, et al. Clinical review: Update on hemodynamic monitoring — a consensus of 16. Crit Care 2011;15:229-238.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
