Selenium and Mercury: A Fishy Tale with Promise
Cardiovascular Disease
Selenium and Mercury: A Fishy Tale with Promise
By Howell Sasser, PhD
Research Review Coordinator, Manila Consulting, McLean, VA; Adjunct Lecturer, Department of Epidemiology and Community Health, New York Medical College, Valhalla, NY
Dr. Sasser reports no financial relationships relevant to this field of study.
It is proverbial in modern life that the health benefits claimed initially for foods and natural products will eventually prove to be overly optimistic (for example, oat bran and dark chocolate), or at least subject to balancing against potential harms (for example, bacterial contamination of fresh produce and unpasteurized juices). Such cautionary examples should not — and usually do not — prevent appropriate use of such items, provided that we have a realistic idea of what they can do and what their attendant harms may be.
A good example of this is high-fat fish and the potential presence of heavy metals. Since at least the 1990s, there has been strong and consistent evidence that the n-3 fatty acids (eicosapentaenoic and docosahexaenoic acid) found in “oily” marine species such as salmon, tuna, and mackerel have significant cardiovascular health benefits. A 1997 meta-analysis showed clinically meaningful reductions in triacylglycerol (triglycerides) with higher fatty fish consumption.1 A 2002 American Heart Association consensus statement added to this, noting n-3 fats’ role in controlling heart rhythm, hemodynamics, and arterial endothelial function.2
The drawback to these positive effects lies in the biology and natural history of the relevant fish species. They are mostly carnivorous and at the top of their respective food chains. Consequently, the dietary and environmental factors of all lower species are concentrated in their diets and in their tissues, a phenomenon called bioaccumulation or biomagnification. This includes heavy metals such as mercury. Mercury typically enters bodies of water from natural or human sources as elemental mercury (Hg) and then is converted by microbial action to the organic compound methylmercury (CH3Hg+, sometimes also written as MeHg+). In this form, it is readily taken up by insects and other small prey species, beginning the process of bioaccumulation.3
By the time some large and long-lived species are caught for human consumption, mercury levels in them can be high enough to pose a meaningful threat to humans, particularly fetuses and small children whose neurological systems are still developing. This has led the U.S. Environmental Protection Agency and Food and Drug Administration to recommend that children and pregnant women restrict their consumption of certain high-fat fish.4 However, it should be noted that mercury levels can vary widely, even within the same species, over time and as a result of the production methods used (i.e., farm-raised vs. wild-caught). (One source for technical data is FDA’s report on surveillance of fish stocks between 1990 and 2010, which is accessible at: www.fda.gov/food/foodsafety/product-specificinformation/seafood/foodbornepathogenscontaminants/methylmercury/ucm115644.htm.) This complexity has created confusion among consumers as to what kinds of fish are safe to include in their diets.
Selenium to the Rescue?
Understandably, there is interest in what strategies may be available to mitigate the danger in what is otherwise a very healthful category of foods. The most obvious approach — curtail at least those environmental emissions of mercury that are of human origin — is enmeshed with political and economic concerns. It is also beyond the control of the individual consumer. Another possible avenue of attack comes from observation of a natural process, ironically best described in fish models.
Selenium (Se) is an elemental substance that forms compounds essential for most plant and animal life.5 In mammals, selenoproteins play a role in thyroid regulation, and selenium is a key component of the antioxidant enzymes glutathione peroxidase and thioredoxin reductase. It has been shown that selenium binds preferentially with mercury to form stable mercury selenide compounds (HgSe).6 This process effectively sequesters methylmercury and prevents it from reaching vulnerable tissues in a form capable of causing damage. It has also been suggested that the antioxidant compounds of which selenium is a part may help to prevent cellular damage by scavenging free radical oxygen species created by biochemical processes involving methylmercury among other sources.7
It should be noted that the chemical processes by which selenium reduces the presence of mercury in the tissue also deplete the body’s supply of selenium. A deficiency of selenium has an independent set of potential health consequences, including thyroid dysregulation, immune system dysfunctions, and infertility.7 A diet high in fatty fish would seem to argue for careful attention to adequate consumption of selenium, both for the protective role it may play with mercury and also precisely because this function may leave inadequate selenium for other important processes. However, there is little experimental evidence in vertebrate mammals, and none in humans, for the magnitude or consistency of the effect of selenium supplementation. Nevertheless, the available evidence is intriguing.
What the Science Tells Us
In areas with high industrial output of mercury (for instance, from factory smokestacks), some bodies of water have come to have such high levels that they have been declared off limits for fishing for human consumption. A group in Sweden experimented with artificially raising the selenium content of one such “black-listed” lake for a period of several years and then monitored fish in the lake for the mercury level in their tissues.8 They showed marked, statistically significant reductions in mercury in several fish species, reaching levels below the “black-list” threshold for as long as the supplementation continued.
A Japanese group fed juvenile rats a diet of fish with a known content of methylmercury, and a variable amount of natural or supplemental selenium.9 They found that selenium in blood, brain, and spinal cord was positively correlated (r between 0.69 and 0.90) with protection from neurological damage attributable to mercury. This finding was partially replicated by another group that studied mercury and antioxidant exposure in gravid rats and their offspring.10 Exposure to mercury during pregnancy reduced the survival rate of the resultant offspring. While nursing, the mothers showed better gains in weight and neurological function when supplemented with a combination of selenium and vitamin E. However, the mothers’ supplementation did not appear to have any beneficial effects in their offspring.
Available evidence in humans to date is entirely observational. A population survey (n = 1216) in Britain collected 7-day-recall food frequency data, from which five categories of fish intake were summarized — fried white fish, other white fish, shellfish, oily fish, and all fish combined.11 Mercury and selenium levels in blood and blood components also were assessed. In linear regression models, consumption of all categories of fish except fried white fish was strongly and positively associated with mercury in whole blood and with selenium in red blood cells and plasma (P < 0.0001 in all cases). Blood mercury was also strongly and positively correlated with red cell and plasma selenium. They concluded from this that fish consumption was the source of both factors. It should be noted, however, that a number of other food categories also showed significant correlations with both mercury and selenium.
A Chinese group sampled body fluids from a group of 72 people living in a mercury-contaminated region.12 Mercury exposure was categorized as high or low, based on serum and urine levels, and selenium was measured in the same samples. They found a statistically significant correlation between mercury and selenium levels in urine (r = 0.625, P < 0.001), but not in serum. These results are cross-sectional only, so no inference about causation is possible. However, there does seem to be evidence that mercury and selenium covary in humans in a predictable way.
Norwegian investigators conducted a retrospective study comparing a group of 20 people who reported having symptoms that could be attributed to their mercury amalgam fillings and 21 people who also had amalgam fillings but did not report relevant symptoms.13 They found that the median serum concentration of selenium in those reporting symptoms was statistically significantly lower than in those not reporting symptoms (119.2 mcg/L vs. 130.3 mcg/L, P = 0.003). A separate comparison of people who had had amalgam fillings removed with others who had never had amalgam fillings showed no difference in serum selenium levels. This finding is suggestive, but does not allow any inference about which factor (presence of fillings, serum mercury, serum selenium) appeared first or what the pattern of causation may be.
Conclusion
The available evidence seems adequate to conclude that there is an interaction between mercury and selenium that may be clinically relevant. Studies of fish and non-human vertebrates show strong correlations between higher selenium levels and lower mercury levels, as well as lower rates of neurological deficits that are attributable to mercury toxicity. There is also suggestive evidence in humans that lower selenium levels may be associated with a greater chance of mercury-associated health problems. However, there is as yet no definitive evidence in humans that consumption of any defined level of selenium, or supplementation with selenium at doses higher than required to meet non-mercury-related physiological needs, protects against damage caused by dietary or environmental mercury exposure.
Given the potential harm arising from consumption of mercury, experimental evidence in a human model is unlikely to be forthcoming. However, it is reasonable to expect that large-scale observational studies and quasi-experimental designs should be possible. Regrettably, there are large populations in many parts of the world living in areas where environmental and/or dietary exposure to mercury makes natural experiments feasible. Further clarification of the mercury/selenium relationship would go a long way toward mitigating a health burden that they bear, and that is unlikely to be remedied at the source in the foreseeable future.
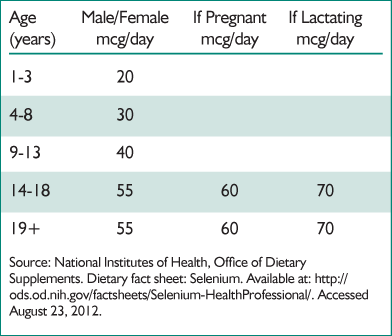
Table 1. Recommended daily allowances of selenium
Recommendations
Selenium is an essential component of the human diet. The recommended daily allowances of selenium, as endorsed by U.S. government agencies, is included in Table 1.
However, consumption of selenium in large quantities can lead to a toxicity syndrome known as selenosis, which is characterized by gastrointestinal distress, fatigue, and irritability, among other symptoms. The Institute of Medicine of the U.S. National Academy of Sciences recommends an upper limit of 400 mcg per day for adults.14 It is important to note that the selenium content in foods can vary dramatically, so consumption of foods known to be rich in selenium (Brazil nuts, as well as plant-based foods from geographic areas with high natural selenium levels) should be managed in light of overall selenium intake.
Although there is no level of mercury recognized as “safe,” the Agency for Toxic Substances and Disease Registry, a component of the Centers for Disease Control and Prevention, has stated that in its opinion a level of CH3Hg+ in fish of no more than 0.003 mg/kg/day represents, “an estimate of the daily human exposure that is likely to be without appreciable risk of adverse non-cancer health effects over a specified duration of exposure.”15
At this point, there does not appear to be adequate evidence to justify recommending that anyone consume extra selenium as a form of mercury prophylaxis. However, consumption of adequate quantities of essential nutrients, including selenium, has proven benefit in a variety of other ways and can be recommended on its own merits. Patients who wish to ensure that they consume adequate quantities of n-3 fatty acids, but are concerned about mercury or other toxicants in fish, can be offered the alternative of taking fish oil capsules, which avoid the problem altogether. In this, as in many such situations, an open discussion of patient preferences and comfort with risk is a good way to clarify the issues, identify potential misunderstandings, and make appropriate recommendations.
References
1. Harris W. n-3 Fatty acids and serum lipoproteins: Human studies. Am J Clin Nutr 1997;65(Suppl):1645S-1654S.
2. Kris-Etherton PM, et al, for the AHA Nutrition Committee. Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Circ 2002;106:2747-2757.
3. Gilbert SG, Grant-Webster KS. Neurobehavioral effects of developmental methylmercury exposure. Environ Health Perspect 1995;103(Suppl 6):135-142.
4. United States Environmental Protection Agency & United States Food and Drug Administration. 2004. What You Need to Know About Mercury in Fish and Shellfish (EPA-823-R-04-005). Available at: www.fda.gov/Food/FoodSafety/Product-SpecificInformation/Seafood/FoodbornePathogensContaminants/Methylmercury/ucm115662.htm. Accessed August 22, 2012.
5. Moghadaszadeh B, Beggs AH. Selenoproteins and their impact on human health through diverse physiological pathways. Physiol 2006;21:307-315.
6. Ralston NVC, Raymond LJ. Dietary selenium’s protective effects against methylmercury toxicity. Toxicol 2010;278:112-123.
7. Rayman MP. The importance of selenium to human health. Lancet 2000;356:233-241.
8. Paulsson K, Lundbergh K. The selenium method for treatment of lakes for elevated levels of mercury in fish. Sci Total Env 1989;87/88:495-507.
9. Ohi G, et al. The protective potency of marine animal meat against the neurotoxicity of methylmercury: Its relationship with the organ distribution of mercury and selenium in the rat. Fd Cosmet Toxicol 1980;18:139-148.
10. Beyrouty P, Chan HM. Co-consumption of selenium and vitamin E altered the reproductive toxicity of methylmercury in rats. Neurotoxicol Teratol 2006;28:49-58.
11. Bates CJ, et al. Dependence of blood indices of selenium and mercury on estimated fish intake in a national survey of British adults. Pub Health Nutr 2007;10:508-517.
12. Chen C, et al. The roles of serum selenium and selenoproteins on mercury toxicity in environmental and occupational exposure. Environ Health Perspect 2006;114:297-301.
13. Høl PJ, et al. Dental amalgam and selenium in blood. Env Res A 2001;87:141-146.
14. National Institutes of Health, Office of Dietary Supplements. Dietary fact sheet: Selenium. Available at: http://ods.od.nih.gov/factsheets/Selenium-HealthProfessional/. Accessed August 23, 2012.
15. Agency for Toxic Substances and Disease Registry. 2011. Minimal Risk Levels (MRLs) for Hazardous Substances. Available at: http://www.atsdr.cdc.gov/mrls/mrllist.asp. Accessed September 10, 2012.
It is proverbial in modern life that the health benefits claimed initially for foods and natural products will eventually prove to be overly optimistic (for example, oat bran and dark chocolate), or at least subject to balancing against potential harms (for example, bacterial contamination of fresh produce and unpasteurized juices). Such cautionary examples should not and usually do not prevent appropriate use of such items, provided that we have a realistic idea of what they can do and what their attendant harms may be.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
