Right Heart Failure in the ICU
By Trushil Shah, MD, MSc
Assistant Professor of Medicine, University of Texas Southwestern Medical Center, Dallas
Dr. Shah reports he is on the speakers bureau of Gilead Sciences.
Right ventricular (RV) failure continues to be a formidable clinical challenge in critical care medicine. Although no consensus definition of RV failure exists, it is defined commonly as the clinical result of RV dysfunction with onset of hypotension or end organ damage.1 Clinically, RV failure is characterized by reduced cardiac output (cardiac index < 2.5 L/min/m2) and elevated right-sided filling pressures (right atrial pressure > 8 mmHg).2 Regardless of the etiology, pulmonary hypertension and RV failure carries a poor prognosis. Prompt identification and treatment is warranted.1,3-6 Unfortunately, there is considerable lack of evidence in the management of RV failure in the medical ICU. Most recommendations are based on expert opinion or low-quality evidence.1,7
PATHOPHYSIOLOGY AND ETIOLOGY OF RV FAILURE
Normal pulmonary circulation is a low-resistance, low-pressure, high-compliance, high-flow system. Generally, RV afterload is low, and the normal thin-walled RV generates significantly lower pressures compared to the left ventricle (LV). The most common causes of RV failure include RV ischemia (e.g., RV infarction) or a sudden increase in RV afterload (e.g., pulmonary embolism, worsening of pulmonary arterial hypertension). If RV afterload increases gradually, the RV can undergo remodeling and adapt, but it adapts poorly to sudden increases in afterload.8,9 A normal RV is unable to acutely increase mean pulmonary pressures > 40 mmHg, whereas a chronically remodeled RV can mount systematic level pressures.8,9
When decreased contractility causes RV failure, as in the case of RV infarction, afterload generally is low, and cardiac output is dependent on preload. However, in many other etiologies of RV failure, there is an acute increase in RV afterload (e.g., acute respiratory distress syndrome, pulmonary embolism, pulmonary arterial hypertension). Timing of RV failure depends on the severity of the increase in afterload and pre-event RV function. For example, patients with severe pulmonary arterial hypertension might exhibit preexisting RV dysfunction and carry little reserve to compensate for acute increases in preload or afterload. Also, the RV and LV are interdependent through the presence of the interventricular septum and pericardium. Normally, LV systolic function helps RV ejection because of this interdependence.10,11 However, in right heart failure, increased RV pressures and pericardial pressures lead to septal wall displacement toward the LV, causing underfilling and LV diastolic dysfunction.10-12 In this case, pulmonary capillary wedge pressure (PCWP) becomes unreliable as an estimate of LV preload. Transmural pressures (PCWP — right atrial pressure) should be used instead.12 Thus, a dilated and overloaded RV can lead to decreased LV filling and decreased cardiac output. Normal right coronary blood flow is dependent on the pressure gradient between the aorta and RV, and occurs during both systole and diastole because of normal low RV pressures. However, in RV failure, increases in RV pressure and oxygen demand from increased contractility may cause RV ischemia, which further worsens RV function.13
Mechanical ventilation can contribute to RV failure by preload reduction due to increased intrathoracic pressures and decreased venous return. It can also increase afterload as a result of alveolar over distention due to high positive end-expiratory pressure (PEEP) and/or high tidal volumes.14 Higher tidal volumes, plateau pressures, and driving pressures also can increase cytokine release, leading to endothelial injury and dysfunction in the pulmonary circulation.14
Similarly, cytokine release in sepsis can induce pulmonary vascular endothelial dysfunction and cause an increase in pulmonary vascular resistance (PVR).15 Hypoxemia, hypercapnia, and acidosis lead to pulmonary vasoconstriction.16 In certain situations, such as during cardiac surgery, cardiopulmonary bypass can induce pulmonary vascular dysfunction and an increase in PVR in addition to lung injury.17
DIAGNOSIS OF RV FAILURE
A high index of suspicion is needed for the diagnosis of RV failure. A history of the underlying cause might be a first clue to diagnosis. Transthoracic echocardiogram (TTE) is a reasonable screening tool to assess severity of right heart failure as well as to rule out left heart failure and valvular heart disease as the cause of pulmonary hypertension. TTE should be used first, prior to more invasive measures like pulmonary artery catheters (PAC).
Although PAC is the gold standard for diagnosing pulmonary hypertension, routine use of a PAC is not recommended for most ICU patients.18 Recommendations against the use of PACs have been more clear in acute lung injury and cardiogenic shock secondary to left heart failure, but these studies excluded patients with group 1 pulmonary hypertension.18
In the latter subset of patients with RV failure, PACs can be used to establish a diagnosis or guide management in select patients in whom TTE is not sufficient for diagnosis or management. In postoperative cardiac surgery patients, it is reasonable to use PACs for postoperative management. However, in these cases, PACs should be removed as soon as their utility is over. Various serum markers of organ dysfunction, like creatinine, liver enzymes, and lactic acid, can be used to assess severity of RV failure and response to therapy.
N-terminal pro-brain natriuretic peptide (NT-proBNP) and BNP are important markers. Elevated levels predict increased mortality.19-21 For all patients with unexplained RV failure, pulmonary embolism should be suspected. Pulmonary embolism is a common cause of RV failure, even in patients who present with preexisting RV dysfunction. CT angiogram should be considered for acute pulmonary embolism, while a ventilation-perfusion scan should be considered if chronic thromboembolism is suspected. A right-sided ECG and troponin levels should be obtained if RV infarction is suspected.
MANAGEMENT OF RV FAILURE
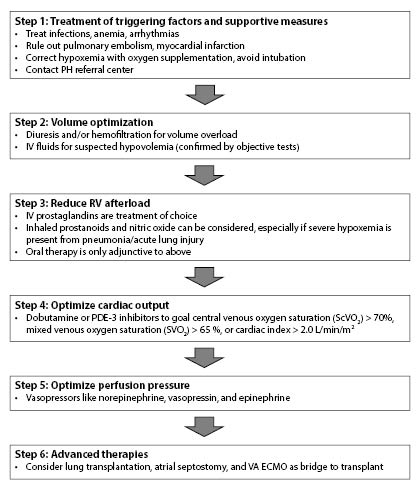
Management of RV failure includes treating the underlying disease, optimizing volume status, providing hemodynamic support, and avoiding further injury to the RV or an increase in afterload. Management of these patients should be handled at an expert pulmonary hypertension center and considered in a stepwise manner.2
Step 1: Treatment of triggering factors and supportive measures. In the current era, RV failure is an uncommon first presentation of pulmonary arterial hypertension (PAH) because of improvement in diagnosis and treatment of this disease. However, from time to time, these cases occur and must be identified and treated promptly. Most of these cases feature a triggering factor that leads to acute decompensation of the patient’s pulmonary hypertension.21 Forty-one percent of patients in a French series experienced a triggering factor, most commonly infection, anemia, arrhythmia, pulmonary embolism, volume overload, or myocardial infarction.21 These factors should be identified and treated promptly.
Infection needs special mention since documented infection was the strongest predictor of death in that study. In addition to usual sources of infection, patients in right heart failure demonstrate increased bacterial translocation from the bowel due to gut edema from elevated central venous pressures, leading to loss of intestinal barrier function.2 PAH patients do not tolerate atrial arrhythmias well because of loss of atrial kick.
A goal should be to restore sinus rhythm with either electric or chemical cardioversion. Rate-controlling medications like calcium channel blockers and beta-blockers should be avoided as they are poorly tolerated in these patients. Anemia is common, mainly because of gastrointestinal (GI) bleeding. To optimize the oxygen-carrying capacity of blood, optimal hemoglobin levels should be maintained. Again, this subpopulation has not been included in most studies; hence, maintaining a hemoglobin level > 8 g/dL is reasonable. Endotracheal intubation and mechanical ventilation should be avoided if at all possible. If absolutely necessary, sedation for anesthesia should be considered carefully as the drop in systemic vascular resistance (SVR) is poorly tolerated in RV failure patients. Because of its minimal cardiovascular effects, etomidate is the induction agent of choice.22
Another option is to use awake fiber optic intubation with local anesthesia. Pretreatment with a vasopressor medication should be considered, too. Post-intubation PEEP, plateau pressures, driving pressures, hypercapnia, hypoxia, and acidosis should be minimized as much as possible.14
Step 2: Volume optimization. Most patients exhibit volume overload and need prompt diuresis and/or hemofiltration. However, a minority demonstrate hypovolemia and might need a small amount of fluid. Most routinely used measures of volume status in the ICU are not studied in this population. More than one test should be used prior to fluid administration. Generally, fluid administration should be in small amounts (e.g., 250 cc normal saline or lactated Ringer’s).
Step 3: Reduce RV afterload. In patients with PAH with RV failure, inhaled or IV prostanoids are the drugs of choice. IV epoprostenol has been shown to offer a mortality benefit in these patients.23 However, these drugs cause a drop in SVR in addition to decreasing PVR. These patients may need administration of vasopressor medications to maintain adequate SVR. For the same reasons, these medications must be started at a lower dose and increased gradually. Oral medications (e.g., PDE-5 inhibitors, endothelin receptor antagonists) generally are not useful in the setting of right heart failure, but can be used as an adjunct to prostanoids.
Step 4: Optimize cardiac output. Inotropes can be used to augment RV contractility. PDE-3 inhibitors (e.g., milrinone) and dobutamine are used commonly. As dobutamine causes more tachyarrhythmias, PDE-3 inhibitors are preferred. Additionally, PDE-3 inhibitors also can induce pulmonary vasodilation and help with afterload reduction.24 Systemic hypotension is a common limitation for using these medications. Concomitant vasopressors often are needed.
Step 5: Optimize perfusion pressure. With the use of pulmonary vasodilators like IV prostanoids and inotropes, systemic hypotension is common; hence, vasopressors are needed commonly. Triggering factors like sepsis and GI bleed also cause a decrease in SVR and may require vasopressor support. Based on current evidence and experience, low-dose vasopressin appears to be beneficial, but higher doses may be harmful.25,26 Vasopressin at 0.01-0.04 U/min can be used to increase SVR. High-dose dopamine should be avoided, as dopamine causes more arrhythmias as compared to norepinephrine and an increased rate of death in a subgroup of patients with left-sided cardiogenic shock.27 Lower-dose dopamine might help with inotropic and chronotropic support, but data to support this are weak. Norepinephrine should be used as the vasopressor of choice. Data to support its use in PAH are weak but are extrapolated from studies in circulatory and cardiogenic shock.27 Epinephrine can be used as the next vasopressor.
Step 6: Advanced therapies. Atrial septostomy can be considered after stabilization of the patient. This helps decrease RV filling pressures and subsequently helps the failing RV by decreasing preload.28 Since this procedure causes a right to left shunt, it is only appropriate for patients with a room air SpO2 > 80%. Lung or heart-lung transplantation remains an important and last treatment option for patients with progressive RV failure despite the above measures. For patients who are transplant candidates, venous arterial extracorporeal membrane oxygenation is used often as a bridge to transplant.29
SUMMARY
Right heart failure in the ICU remains an important challenge. Prompt identification and treatment of the underlying disease is warranted. Additionally, most patients with RV failure need volume optimization and inotropic and vasopressor support, too. In patients with PAH, a stepwise approach to treatment of RV failure should be adopted with special effort to identify and treat triggering factors.
Figure 1. Treatment Algorithm for Right Ventricular Failure in the ICU |
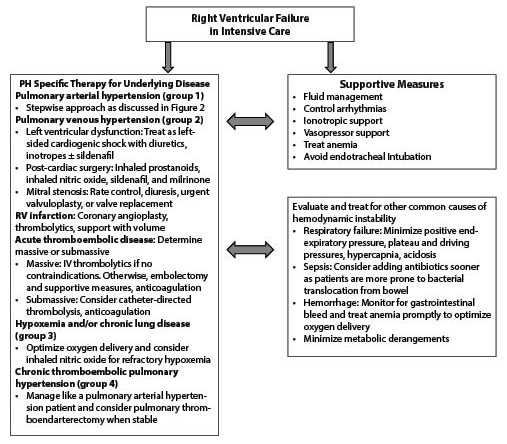 |
Figure 2. Stepwise Approach to Right Ventricular Failure in Pulmonary Arterial Hypertension Patients |
|
Step 1 and 2 are needed in every patient. Escalation to the next step is recommended if not responding adequately. |
 |
REFERENCES
- Price LC, et al. Pulmonary vascular and right ventricular dysfunction in adult critical care: Current and emerging options for management: A systematic literature review. Crit Care 2010;14:R169.
- Hoeper MM, Granton J. Intensive care unit management of patients with severe pulmonary hypertension and right heart failure. Am J Respir Crit Care Med 2011;184:1114-1124.
- Osman D, et al. Incidence and prognostic value of right ventricular failure in acute respiratory distress syndrome. Intensive Care Med 2009;35:69-76.
- Jacobs AK, et al. Cardiogenic shock caused by right ventricular infarction: A report from the SHOCK registry. J Am Coll Cardiol 2003;41:1273-1279.
- Vieillard-Baron A, et al. Acute cor pulmonale in massive pulmonary embolism: Incidence, echocardiographic pattern, clinical implications and recovery rate. Intensive Care Med 2001;27:1481-1486.
- Vieillard-Baron A, et al. Acute cor pulmonale in acute respiratory distress syndrome submitted to protective ventilation: Incidence, clinical implications, and prognosis. Crit Care Med 2001;29:1551-1555.
- Delcroix M, Naeije R. Optimising the management of pulmonary arterial hypertension patients: Emergency treatments. Eur Respir Rev 2010;19:204-211.
- Chen EP, et al. Molecular and functional mechanisms of right ventricular adaptation in chronic pulmonary hypertension. Ann Thorac Surg 1999;67:1053-1058.
- Chin KM, et al. The right ventricle in pulmonary hypertension. Coron Artery Dis 2005;16:13-18.
- Gan C, et al. Impaired left ventricular filling due to right-to-left ventricular interaction in patients with pulmonary arterial hypertension. Am J Physiol Heart Circ Physiol 2006;290:H1528-H1533.
- Vonk-Noordegraaf A, et al. Interventricular mechanical asynchrony due to right ventricular pressure overload in pulmonary hypertension plays an important role in impaired left ventricular filling. Chest 2005;128:628S-630S.
- Belenkie I, et al. Ventricular interaction: From bench to bedside. Ann Med 2001;33:236-241.
- Vlahakes GJ, et al. The pathophysiology of failure in acute right ventricular hypertension: Hemodynamic and biochemical correlations. Circulation 1981;63:87-95.
- Vieillard-Baron A, Jardin F. Why protect the right ventricle in patients with acute respiratory distress syndrome? Curr Opin Crit Care 2003;9:15-21.
- Chan CM, Klinger JR. The right ventricle in sepsis. Clin Chest Med 2008;29:661-676.
- Bindslev L, et al. Hypoxic pulmonary vasoconstriction in man: Effects of hyperventilation. Acta Anaesthesiol Scand 1985;29:547-551.
- Ng CS, et al. Pulmonary dysfunction after cardiac surgery. Chest 2002;121:1269-1277.
- Wiedemann H, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med 2006;354:2564-2575.
- Nickel N, et al. The prognostic impact of follow-up assessments in patients with idiopathic pulmonary arterial hypertension. Eur Respir J 2012;39:589-596.
- Leuchte HH, et al. Characterization of brain natriuretic peptide in long-term follow-up of pulmonary arterial hypertension. Chest 2005;128:2368-2374.
- Sztrymf B, et al. Prognostic factors of acute heart failure in patients with pulmonary arterial hypertension. Eur Respir J 2010;35:1286-1293.
- Pritts CD, Pearl RG. Anesthesia for patients with pulmonary hypertension. Curr Opin Anaesthesiol 2010;23:411-416.
- Barst RJ, et al. A comparison of continuous intravenous epoprostenol (prostacyclin) with conventional therapy for primary pulmonary hypertension. N Engl J Med 1996;334:296-301.
- Chen EP, et al. Milrinone improves pulmonary hemodynamics and right ventricular function in chronic pulmonary hypertension. Ann Thorac Surg 1997;63:814-821.
- Evora PR, et al. Arginine vasopressin induces endothelium-dependent vasodilatation of the pulmonary artery. V1-receptor-mediated production of nitric oxide. Chest 1993;103:1241-1245.
- Leather HA, et al. Effects of vasopressin on right ventricular function in an experimental model of acute pulmonary hypertension. Crit Care Med 2002;30:2548-2552.
- De Backer D, et al. Comparison of dopamine and norepinephrine in the treatment of shock. N Engl J Med 2010;362:779-789.
- Sandoval J, et al. Effect of atrial septostomy on the survival of patients with severe pulmonary arterial hypertension. Eur Respir J 2011;38:1343-1348.
- Olsson KM, et al. Extracorporeal membrane oxygenation in nonintubated patients as bridge to lung transplantation. Am J Transplant 2010;10:2173-2178.
Right ventricular (RV) failure continues to be a formidable clinical challenge in critical care medicine. Regardless of the etiology, pulmonary hypertension and RV failure carries a poor prognosis. Prompt identification and treatment is warranted. Unfortunately, there is considerable lack of evidence in the management of RV failure in the medical ICU. Most recommendations are based on expert opinion or low-quality evidence.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
