The Epidemic of Vaping-Associated Lung Injury
By Trushil Shah, MD, MSc
Assistant Professor of Medicine, University of Texas Southwestern, Dallas
Dr. Shah reports that he has received grant/research support from Actelion Pharmaceuticals, Bayer Pharmaceuticals, and Liquidia Technologies.
Since the introduction of e-cigarettes in 2004, there has been an exponential increase in their use.1 While initially intended to be used for smoking cessation, an increasing number of nonsmokers, many of whom are adolescents, use e-cigarettes.2,3 In the most basic form, these devices consist of a battery that powers a heating coil when activated. This heating coil then transfers heat to a wick soaked in “vaping juice,” which converts the liquid into vapor for direct inhalation.1 Now more commonly known as electronic nicotine delivery systems (ENDS), many of these devices contain other substances like tetrahydrocannabinol (THC) and cannabidiol (CBD), with or without nicotine. These devices come in different forms that could look like everyday objects, such as USB flash drives, credit cards, highlighters, or pens. They also come in different flavors.3 Because of the availability of flavors and the ease of disguising these devices, their use has become popular among teenagers and youth.3 In one survey of high school and middle school students, 60% used a brand called JUUL, which is well known for making flavored, USB-shaped vaping devices.3
Few case reports of vaping-associated lung injury were reported before 2019.4,5 Since June 2019, there has been a considerable rise in reported cases of vaping-associated lung injury.6-10 As of Nov. 20, 2019, 2,290 cases of e-cigarette or vaping- associated lung injury (EVALI) have been reported to the Centers for Disease Control and Prevention (CDC).11 In the same time frame, 47 deaths from EVALI have been confirmed across 25 states and the District of Columbia.11 Because of the heterogeneous nature of lung injury in EVALI and possible under-reporting, the actual burden of disease may be much higher.6 With symptoms indistinguishable from influenza, a high index of suspicion is essential to the diagnosis and treatment of EVALI.
Definition and Case Identification
E-cigarette or vaping-associated lung injury (EVALI) is a diagnosis of exclusion and should be made by ruling out more common causes of acute lung injury, especially infections. The CDC currently uses surveillance definitions of “confirmed” and “probable.”12 A confirmed case is defined as a reported history of e-cigarette/vaping device use within 90 days prior to symptom onset, the presence of pulmonary infiltrates on chest X-ray or ground glass opacities on chest computed tomography (CT), negative influenza PCR, and no evidence of infection or other plausible diagnosis (e.g., cardiac, rheumatologic, neoplastic etiologies, etc.).12 A case is reported as “probable” if there is evidence of infection, or minimum criteria to rule out infection are not met, but the clinical team believes the infection is not the sole cause of lung injury.12
Symptoms and Signs
According to the CDC’s latest report in November 2019, 85% of patients with EVALI present with respiratory symptoms (cough, shortness of breath, chest pain), 76% present with constitutional symptoms (fever, chills, weight loss), and 57% report gastrointestinal symptoms (nausea, vomiting, abdominal pain, diarrhea).6 Interestingly, very few patients presented with only respiratory symptoms (9%), and most patients had a combination of respiratory, constitutional, and gastrointestinal symptoms.6,13 Since these symptoms are very nonspecific, influenza and other viral infections should be ruled out before diagnosing EVALI. Hypoxemia is the most common sign, and the degree of hypoxemia may vary from mild to acute hypoxemic respiratory failure.13,14 Fever, tachycardia, leukocytosis, elevated erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and procalcitonin levels are seen commonly.14
Imaging and Pathology Findings
The most common chest X-ray findings are bilateral infiltrates. In most cases, CT chest imaging reveals bilateral ground glass opacities with subpleural sparing and septal thickening. However, cases of pleural effusion, recurrent pneumothorax, and pneumomediastinum have been reported as well.15,16 Various different pathological and imaging presentations have been described, including acute hypersensitivity pneumonitis, acute eosinophilic pneumonia, diffuse alveolar hemorrhage, lipoid pneumonia, respiratory bronchiolitis-interstitial lung disease, organizing pneumonia, and acute respiratory distress syndrome (ARDS).16 Pathology from a more recent epidemic has been described as mainly acute lung injury/diffuse alveolar damage and organizing pneumonia as well as findings of foamy macrophages and pneumocyte vacuolization.17,18 In addition to ruling out infection, bronchoalveolar lavage could help diagnose acute eosinophilic pneumonia, acute hypersensitivity pneumonitis, diffuse alveolar hemorrhage, and lipoid pneumonia.16
Possible Pathologic Mechanisms
Many studies have shown the harmful effects of vaping affecting the respiratory epithelium, lung endothelium, and nasal epithelium, resulting in decreased gas exchange and an increase in matrix metalloproteinases and neutrophil elastases.19-21 However, current data and experience with e-cigarettes/vaping are insufficient to predict long-term toxicities.21 The recent outbreak of vaping-related lung injury seems to differ from the traditional toxicity of e-cigarettes in that it causes an acute lung injury pattern, which presents within days to weeks of exposure. In their investigation of samples collected from patients with EVALI, the CDC and Food and Drug Administration (FDA) found that most patients with lung injury had used a THC-based product or a combination of nicotine and THC. Based on a CDC study, 85% of patients with EVALI reported THC-containing e-cigarette use as opposed to 21% of survey respondents without EVALI.9 A higher proportion of patients with EVALI also were found to use THC-containing vapes from an informal source (odds ratio [OR] = 9.2) and more often used Dank Vapes (i.e., cheaper, counterfeit brands from unreliable sources) (OR = 8.5).9 Among bronchoalveolar lavage samples from patients with EVALI, the CDC found vitamin E acetate in all (29/29) samples, THC metabolites in 23/28 samples, and nicotine in 16/26 samples.22 Vitamin E acetate is used as an additive or thickening agent in THC-containing e-cigarettes and vaping products.22 However, the presence of vitamin E acetate only establishes an association and does not imply causality.
Treatment and Management
Most patients with EVALI present with clinical and imaging findings similar to ARDS.6,23 All patients should be screened for influenza and other infections.23 While no definitive treatment of EVALI is currently available, the majority of patients were treated with corticosteroids and demonstrated improvement.14,23,24 However, cases of recovery without corticosteroid therapy also have been reported.14 If corticosteroids are used, prednisone 1 mg/kg or the equivalent seems to be an appropriate starting dose, but this can be individualized based on other patient factors.14,24 Empiric antibiotics and antivirals should be considered in patients with suspected infection.23 Bronchoalveolar lavage and transbronchial biopsy may be helpful to rule out other causes, such as infection, acute eosinophilic pneumonia, acute hypersensitivity pneumonitis, lipoid pneumonia, organizing pneumonia, and diffuse alveolar hemorrhage.23 In ventilated patients, ARDS management protocols, such as low tidal volume ventilation, restrictive fluid strategy, etc., should be used.
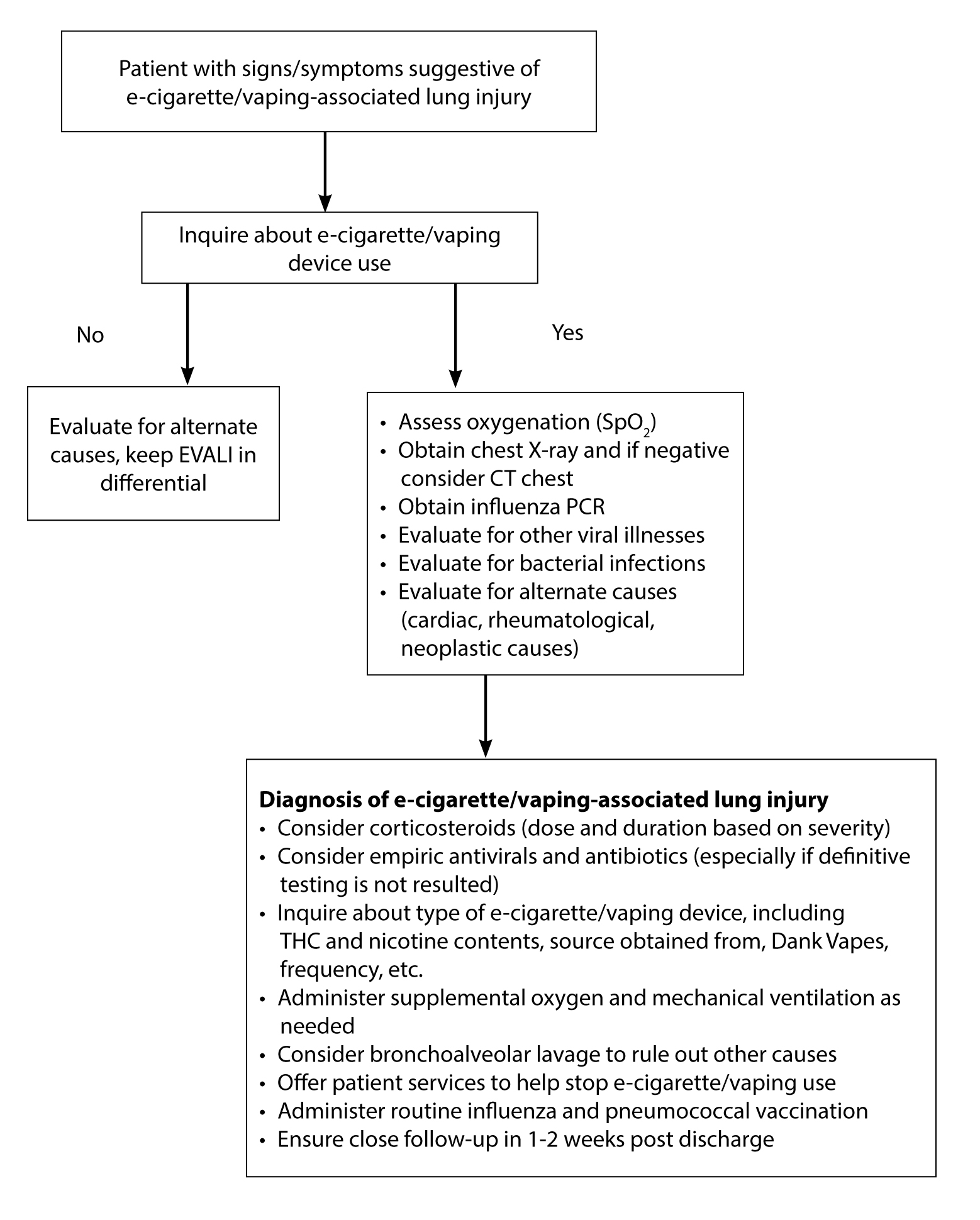
The proportion of patients developing pneumothorax and pneumomediastinum seems to be higher in EVALI patients.24 After signs of improvement, close follow-up is important, since cases of recurrence have been described, especially in patients who restart vaping/e-cigarettes.23 In patients presenting with symptoms of EVALI without clear chest X-ray findings, CT chest should be considered because cases have been described in which the chest X-ray has been reportedly normal.23 An algorithm for managing inpatients with EVALI is summarized in Figure 1. If such patients are seen in an outpatient setting or are discharged after observation, close outpatient follow-up in 48 hours should be arranged because cases of acute worsening within that time frame have been described.23 All patients should be advised against using e-cigarettes/vaping products, especially those containing THC.23 Patients with EVALI should be strongly advised not to return to e-cigarette/vaping and to come back for evaluation if symptoms recur.23
Figure 1: Algorithm for Inpatient Management of E-Cigarette/Vaping-Associated Lung Injury Patients |
 |
Conclusion
E-cigarette/vaping-associated lung injury cases have risen sharply in the past six months, and they are anticipated to continue to increase in the near future. A high index of suspicion is needed to diagnose and treat patients with EVALI. THC and vitamin E-containing products seem to be associated with the current epidemic. Investigations are ongoing to demystify the current EVALI epidemic further.
REFERENCES
- Collaco JM, Drummond MB, McGrath-Morrow SA. Electronic cigarette use and exposure in the pediatric population. JAMA Pediatr 2015;169:177-182.
- McMillen RC, Gottlieb MA, Winickoff JP. E-cigarettes — The roles of regulation and clinicians. JAMA Intern Med 2015;175:1603-1604.
- Cullen KA, Gentzke AS, Sawdey MD, et al. E-cigarette use among youth in the United States, 2019. JAMA 2019 Nov. 5. [Epub ahead of print].
- Viswam D, Trotter S, Burge PS, Walters GI. Respiratory failure caused by lipoid pneumonia from vaping e-cigarettes. BMJ Case Rep 2018;2018.
- Sommerfeld CG, Weiner DJ, Nowalk A, Larkin A. Hypersensitivity pneumonitis and acute respiratory distress syndrome from e-cigarette use. Pediatrics 2018;141(6).
- Chatham-Stephens K, Roguski K, Jang Y, et al. Characteristics of hospitalized and nonhospitalized patients in a nationwide outbreak of e-cigarette, or vaping, product use-associated lung injury - United States, November 2019. MMWR Morb Mortal Wkly Rep 2019;68:1076-1080.
- Davidson K, Brancato A, Heetderks P, et al. Outbreak of electronic-cigarette-associated acute lipoid pneumonia - North Carolina, July-August 2019. MMWR Morb Mortal Wkly Rep 2019;68:784-786.
- Lewis N, McCaffrey K, Sage K, et al. E-cigarette use, or vaping, practices and characteristics among persons with associated lung injury - Utah, April-October 2019. MMWR Morb Mortal Wkly Rep 2019;68:953-956.
- Navon L, Jones CM, Ghinai I, et al. Risk factors for e-cigarette, or vaping, product use-associated lung injury (EVALI) among adults who use e-cigarette, or vaping, products - Illinois, July-October 2019. MMWR Morb Mortal Wkly Rep 2019;68:1034-1039.
- Taylor J, Wiens T, Peterson J, et al; Lung Injury Response Task Force. Characteristics of e-cigarette, or vaping, products used by patients with associated lung injury and products seized by law enforcement - Minnesota, 2018 and 2019. MMWR Morb Mortal Wkly Rep 2019;68:1096-1100.
- Centers for Disease Control and Prevention. Outbreak of lung injury associated with the use of e-cigarette, or vaping, products. Available at: https://www.cdc.gov/tobacco/basic_information/e-cigarettes/severe-lung-disease.html. Accessed Dec. 2, 2019.
- Centers for Disease Control and Prevention. 2019 lung injury surveillance primary case definitions. Sept. 18, 2019. Available at: https://www.cdc.gov/tobacco/basic_information/e-cigarettes/assets/2019-Lung-Injury-Surveillance-Case-Definition-508.pdf. Accessed Dec. 5, 2019.
- Layden JE, Ghinai I, Pray I, et al. Pulmonary illness related to e-cigarette use in Illinois and Wisconsin - Preliminary Report. N Engl J Med 2019 Sep. 6. [Epub ahead of print].
- Kalininskiy A, Bach CT, Nacca NE, et al. E-cigarette, or vaping, product use associated lung injury (EVALI): Case series and diagnostic approach. Lancet Respir Med 2019;7:1017-1026.
- Bonilla A, Blair AJ, Alamro SM, et al. Recurrent spontaneous pneumothoraces and vaping in an 18-year-old man: A case report and review of the literature. J Med Case Rep 2019;13:283.
- Fonseca Fuentes X, Kashyap R, Hays JT, et al. VpALI-vaping-related acute lung injury: A new killer around the block. Mayo Clin Proc 2019; Nov. 20. [Epub ahead of print].
- Butt YM, Smith ML, Tazelaar HD, et al. Pathology of vaping-associated lung injury. N Engl J Med 2019;381:1780-1781.
- Mukhopadhyay S, Mehrad M, Dammert P, et al. Lung biopsy findings in severe pulmonary illness associated with e-cigarette use (vaping): A report of eight cases. Am J Clin Pathol 2019 Oct. 17. [Epub ahead of print].
- Ghosh A, Coakley RD, Ghio AJ, et al. Chronic e-cigarette use increases neutrophil elastase and matrix metalloprotease levels in the lung. Am J Respir Crit Care Med 2019;200:1392-1401.
- Chaumont M, van de Borne P, Bernard A, et al. Fourth generation e-cigarette vaping induces transient lung inflammation and gas exchange disturbances: Results from two randomized clinical trials. Am J Physiol Lung Cell Mol Physiol 2019;316:L705-L719.
- Gotts JE, Jordt SE, McConnell R, Tarran R. What are the respiratory effects of e-cigarettes? BMJ 2019;366:l5275.
- Blount BC, Karwowski MP, Morel-Espinosa M, et al. Evaluation of bronchoalveolar lavage fluid from patients in an outbreak of e-cigarette, or vaping, product use-associated lung injury - 10 states, August-October 2019. MMWR Morb Mortal Wkly Rep 2019;68:1040-1041.
- Jatlaoui TC, Wiltz JL, Kabbani S, et al. Update: Interim guidance for health care providers for managing patients with suspected e-cigarette, or vaping, product use-associated lung injury - United States, November 2019. MMWR Morb Mortal Wkly Rep 2019;68:1081-1086.
- Blagev DP, Harris D, Dunn AC, et al. Clinical presentation, treatment, and short-term outcomes of lung injury associated with e-cigarettes or vaping: A prospective observational cohort study. Lancet 2019 Nov. 8. [Epub ahead of print].
Since June 2019, there has been a considerable rise in reported cases of vaping-associated lung injury.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
